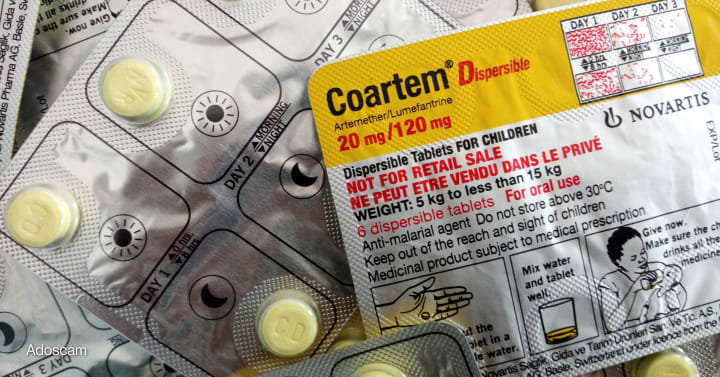
Malaria medicine for infants approved by Swissmedic! Coartem Baby becomes the first antimalarial drug for newborns, targeting babies weighing 2–5 kg. Essential update for government exam aspirants.
🌍 Switzerland Clears First Malaria Medicine for Small Babies
Overview of the Breakthrough
On July 9, 2025, Swissmedic, Switzerland’s medical regulatory authority, granted approval for Coartem Baby (also known as Riamet Baby), designed specifically for infants weighing between 2 kg and 5 kg Developed through a collaboration between Novartis and the Medicines for Malaria Venture (MMV), this sweet-cherry flavored, dissolvable formulation of artemether-lumefantrine fills a critical treatment gap for newborns and very young infants, who were previously excluded from licensed malaria therapies
Designed for Tiny Lives
Prior to this approval, infants weighing under ~4.5 kg had no dedicated antimalarial treatment. Caregivers were forced to adapt higher-dose tablets for older children—risking overdose and ineffective dosing Coartem Baby is specially calibrated for neonates, with a formulation that dissolves in breast milk or water, ensuring accurate dosing and easy administration
Clinical Importance & Roll‑out
This approval comes under Swissmedic’s fast‑track Global Health Products (MAGHP) program, endorsed by the World Health Organization Eight malaria‑endemic African nations—Burkina Faso, Côte d’Ivoire, Kenya, Malawi, Mozambique, Nigeria, Tanzania, and Uganda—have already agreed to accelerated approval within 90 days, paving the way for prompt deployment
Addressing Child Mortality
Malaria remains one of the top killer diseases among children, with nearly 600,000 deaths in 2023, 76% of which occurred in children under five, especially in AfricaThis new pediatric formulation significantly enhances treatment equity and promises to reduce mortality in the most vulnerable age group
Support Beyond Drugs
Novartis commits to distributing Coartem Baby on a largely not-for-profit basis in afflicted regions However, experts emphasize that sustainable access will also depend on transparent pricing, adequate funding, and integration into national malaria control programs, particularly as global aid fluctuates .
.jpg)
📌 Why This News Matters
Protection for the Most Vulnerable
Neonates and low-weight infants are especially prone to severe malaria and mortality, yet lacked dosage-appropriate medication. Now, they have a formulated, clinically tested* treatment designed just for them .
Closing a Vital Treatment Gap
Until now, clinicians relied on “diluted” or improvised dosing of older-child tablets—an unsafe and suboptimal method. Coartem Baby solves this long-standing public health challenge
Aligns with Global Health Goals
This milestone supports WHO’s objectives to reduce malaria-related deaths and amplify access to lifesaving treatments for all age groups, reinforcing commitments toward SDG 3: Good Health and Well‑being.
Boosts Confidence in Pediatric Medicine
The news highlights successful drug formulation for babies, encouraging innovation in pediatric pharmacology and potentially inspiring similar solutions for other infant diseases.
Valuable for Exam Preparation
For competitive exams like PSC, Civil Services, Banking, Railways, this story is a prime example of current affairs aligned with Health & International Relations syllabus—it showcases global cooperation, SDG alignment, and pharmaceutical innovation.
🕰 Historical Context
Past Treatment Landscape
Since Coartem’s debut in 1999, Novartis has provided over 1.1 billion antimalarial courses, primarily for children weighing above 5 kg However, infants were excluded from early clinical trials due to immature metabolism and liver function, necessitating new dosage formulations
Neglect of Neonatal Malaria
There was a misconception that infants were less susceptible due to maternal immunity. However, rising evidence shows 3–18% infection rates even in infants under six months, necessitating targeted drug development .
Rising Challenges
Malaria control has faced setbacks due to global funding cuts and expanding transmission zones driven by climate change, challenging progress in malaria-endemic zonesApproval of Coartem Baby marks a new strategic play in overcoming such hurdles.
Aiding Global Roll‑out
Leveraging the MAGHP fast-track mechanism with WHO aligns with historical efforts to accelerate access to vital drugs in low-income countries, such as HIV/AIDS and tuberculosis medications, demonstrating strengthened commitment.
Key Takeaways from Coartem Baby Approval
| S.No | Key Takeaway |
|---|---|
| 1 | Coartem Baby is the first antimalarial approved for infants weighing 2–5 kg. |
| 2 | It contains artemether-lumefantrine, adjusted dose with dissolvable, cherry-flavored form for easy administration. |
| 3 | Approved by Swissmedic under MAGHP fast-track, with support from WHO. |
| 4 | Eight African countries (e.g., Nigeria, Kenya) fast-tracked approval, streamlining rollout in malarial zones. |
| 5 | Novartis will distribute it mostly on a not-for-profit basis, boosting access amid global health funding uncertainties. |
FAQs: Frequently Asked Questions
1. What is Coartem Baby?
Coartem Baby is a pediatric formulation of the antimalarial drug artemether-lumefantrine, approved for treating malaria in newborns and infants weighing between 2–5 kg.
2. Why was Coartem Baby developed?
It was developed to close the treatment gap for neonates who previously had no approved and accurate dosage for malaria, which is often deadly in this age group.
3. Who approved Coartem Baby for use?
Swissmedic, Switzerland’s national medical regulator, granted approval under the MAGHP (Medicines for Global Health Products) pathway in partnership with WHO.
4. Which pharmaceutical company developed Coartem Baby?
The drug was developed by Novartis in collaboration with the Medicines for Malaria Venture (MMV).
5. Which countries will adopt Coartem Baby next?
Eight malaria-endemic African nations—Burkina Faso, Côte d’Ivoire, Kenya, Malawi, Mozambique, Nigeria, Tanzania, and Uganda—have agreed to approve it within 90 days.
6. What is the formulation of Coartem Baby like?
It is a sweet cherry–flavored dispersible tablet that can dissolve in water or breast milk, making it easier for infants to ingest.
7. What is the significance of MAGHP in this approval?
MAGHP is a WHO-backed fast-track process for getting medicines approved for global health emergencies, especially for low-income countries.
8. Why is malaria particularly dangerous for babies?
Infants have underdeveloped immune systems and are highly vulnerable to severe complications and death due to malaria.
9. How many children died due to malaria in 2023?
According to WHO data, around 600,000 people died due to malaria in 2023, with 76% of them being children under the age of five.
10. Will the drug be affordable?
Yes, Novartis plans to distribute Coartem Baby on a largely not-for-profit basis in malaria-affected regions to improve accessibility.
Some Important Current Affairs Links